Hand Warmer Challenge
Purpose:
Create a safe, inexpensive, cost effective, and efficient hand warmer that you can sell in the student store on campus to make money for the school.
Create a safe, inexpensive, cost effective, and efficient hand warmer that you can sell in the student store on campus to make money for the school.
Beginning of the project:
We first started this project by testing how salts react with water and how much the temperature changed. From this we found that Lithium Chloride or LiCl made the biggest temp. change over the shortest amount of time.
We first started this project by testing how salts react with water and how much the temperature changed. From this we found that Lithium Chloride or LiCl made the biggest temp. change over the shortest amount of time.
Our Ideas:
We had about 7 different main ideas
They all had the same basic ideas but were changed slightly
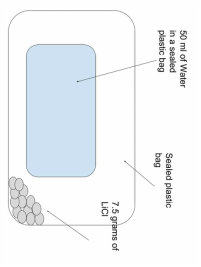
Our first idea was to have a small bag of water in a big bag with the salt, this stayed constant for almost the whole project.
Another idea we had was to use micropipet tubes to slowly let the salt mix, but the were hard to open.
we had different designs like putting pictures on them or different shapes. We found that the rectangle shapes popped the least, so we just stuck with those.
We had about 7 different main ideas
They all had the same basic ideas but were changed slightly
Our first idea was to have a small bag of water in a big bag with the salt, this stayed constant for almost the whole project.
Another idea we had was to use micropipet tubes to slowly let the salt mix, but the were hard to open.
we had different designs like putting pictures on them or different shapes. We found that the rectangle shapes popped the least, so we just stuck with those.
Final Product:
Materials:
5 grams of LiCl
5 grams of CaCl
50 ml of water in a small bag and that inside
A heat sealed double bag
1 ft of Duct tape
Our final cost was about 78 cents and we would sell them for $1.25
Our design:
Our final design was very similar to our original design where it had two bags one holding water inside the one holding the salts. It was different from other groups because we used both Lithium Chloride and Calcium Chloride, the LiCl started the reaction and made a big temperature change, the CaCl works slower and it helped sustain the heat for a longer period of time. Our hand warmer lasted for about 20 mins (this blew a lot of groups out of the water) because of the method of using 2 salts. We also decided to cover the hand warmer in duct tape because it made it less likely for the bag to leak, also the duct tape gave ascetic appeal.
Materials:
5 grams of LiCl
5 grams of CaCl
50 ml of water in a small bag and that inside
A heat sealed double bag
1 ft of Duct tape
Our final cost was about 78 cents and we would sell them for $1.25
Our design:
Our final design was very similar to our original design where it had two bags one holding water inside the one holding the salts. It was different from other groups because we used both Lithium Chloride and Calcium Chloride, the LiCl started the reaction and made a big temperature change, the CaCl works slower and it helped sustain the heat for a longer period of time. Our hand warmer lasted for about 20 mins (this blew a lot of groups out of the water) because of the method of using 2 salts. We also decided to cover the hand warmer in duct tape because it made it less likely for the bag to leak, also the duct tape gave ascetic appeal.
Reflection:
This project was alright, it was interesting to learn about the salt reactions, but I didn't like the project as a whole. It was very simple and was not as interesting in my point of view. I did however, have a great group. We worked very well together and we got along quite well. I did like how we had judges grade our presentation, I thought it was fun. Overall I thought the project was a little lackluster but I did learn a lot which is good.
This project was alright, it was interesting to learn about the salt reactions, but I didn't like the project as a whole. It was very simple and was not as interesting in my point of view. I did however, have a great group. We worked very well together and we got along quite well. I did like how we had judges grade our presentation, I thought it was fun. Overall I thought the project was a little lackluster but I did learn a lot which is good.