Chemical DOMINOES
Purpose: We were supposed to make a machine that showed 3 chemical reactions.
Our plan: Create a Rube Goldberg machine to show our reactions
Our plan: Create a Rube Goldberg machine to show our reactions
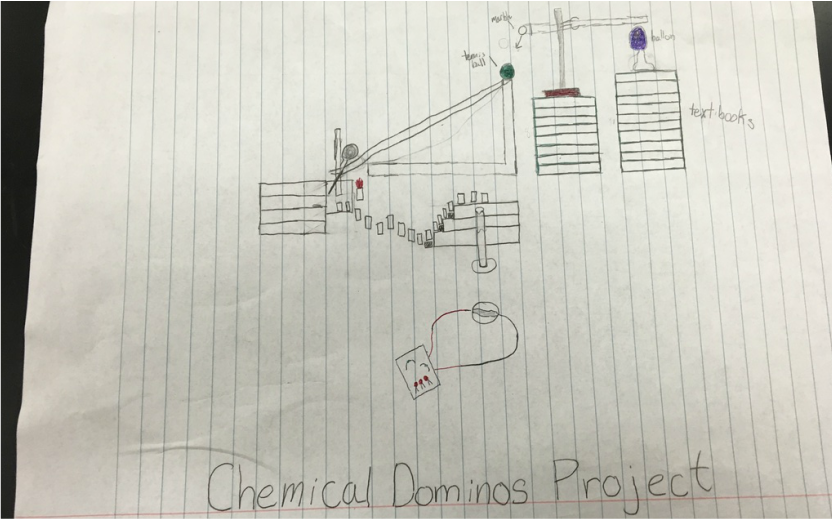
Our Final Project: It had 3 reactions the first reaction started off the machine.
1. We put 50ml of vinegar in a flask, the we filled a balloon with 15g of baking soda. Then we put the balloon on the flask and emptied the baking soda into it.
2. Then the balloon raised a tube that held a marble.
3. Then the marble rolled to hit a tennis ball that rolled down a track
4. As the tennis ball rolled down the ramp it hit a ring stand
5. The ring stand pivoted, knocked over dominoes, and put the magnesium ribbon over the flame.
6. As the Mg Ribbon burns, the dominoes knock over and knocks over a vial holding the copper chloride onto the aluminum connected to the circuit.
7. As the Cu Chloride breaks down the Mg Ribbon should be reacting.
8. After the Mg Ribbon reacts, the copper chloride will break the aluminum foil and light the LED's.
1. We put 50ml of vinegar in a flask, the we filled a balloon with 15g of baking soda. Then we put the balloon on the flask and emptied the baking soda into it.
2. Then the balloon raised a tube that held a marble.
3. Then the marble rolled to hit a tennis ball that rolled down a track
4. As the tennis ball rolled down the ramp it hit a ring stand
5. The ring stand pivoted, knocked over dominoes, and put the magnesium ribbon over the flame.
6. As the Mg Ribbon burns, the dominoes knock over and knocks over a vial holding the copper chloride onto the aluminum connected to the circuit.
7. As the Cu Chloride breaks down the Mg Ribbon should be reacting.
8. After the Mg Ribbon reacts, the copper chloride will break the aluminum foil and light the LED's.
Reactions:
-Baking Soda and Vinegar a.k.a. Sodium Bicarbonate and Acetic Acid
HC2H3O2(aq) + NaHCO3(aq) NaC2H3O2(aq) + H2CO3(aq)
This was a double displacement reaction
-Magnesium Ribbon and Fire
2Mg + O2 ---> 2MgO + Energy
This was a synthesis reaction
-Chopper Chloride and Aluminum foil
2Al + 3CuCl2 ---> 3Cu + 2AlCl3
This was a single displacement reaction
-Baking Soda and Vinegar a.k.a. Sodium Bicarbonate and Acetic Acid
HC2H3O2(aq) + NaHCO3(aq) NaC2H3O2(aq) + H2CO3(aq)
This was a double displacement reaction
-Magnesium Ribbon and Fire
2Mg + O2 ---> 2MgO + Energy
This was a synthesis reaction
-Chopper Chloride and Aluminum foil
2Al + 3CuCl2 ---> 3Cu + 2AlCl3
This was a single displacement reaction
What we learned:
- We learned about the different types of reaction
- How different chemicals can cause different things
- Also how to apply chemistry to physics
Reflection:
Overall this was a great project and my group and I really had fun with it. This project was so cool because it was a throw back to freshman year. Also we got to apply physics to a chemistry project which made me so happy. This project wasn't just making a machine. We did a lot of experiments to find steps for our project before we even started building. We also had to learn about different chemical reactions, those being: single displacement, double displacement, synthesis, decomposition, and combustion reactions. Overall my group was amazing, we had a lot of fun and completed our work in a timely manner. Our project was awesome, it worked a lot of the time, and it was super cool. But it took a lot of time to perfect, and the last step took a couple of minutes to work. We should have created a more concentrated solution so the aluminum could have broken down faster. Overall, this was by far the best project this year.
Overall this was a great project and my group and I really had fun with it. This project was so cool because it was a throw back to freshman year. Also we got to apply physics to a chemistry project which made me so happy. This project wasn't just making a machine. We did a lot of experiments to find steps for our project before we even started building. We also had to learn about different chemical reactions, those being: single displacement, double displacement, synthesis, decomposition, and combustion reactions. Overall my group was amazing, we had a lot of fun and completed our work in a timely manner. Our project was awesome, it worked a lot of the time, and it was super cool. But it took a lot of time to perfect, and the last step took a couple of minutes to work. We should have created a more concentrated solution so the aluminum could have broken down faster. Overall, this was by far the best project this year.
There were some technical difficulties so, I am not able to post the video at this time